Volume 40 Number 2 | April 2026
Summary
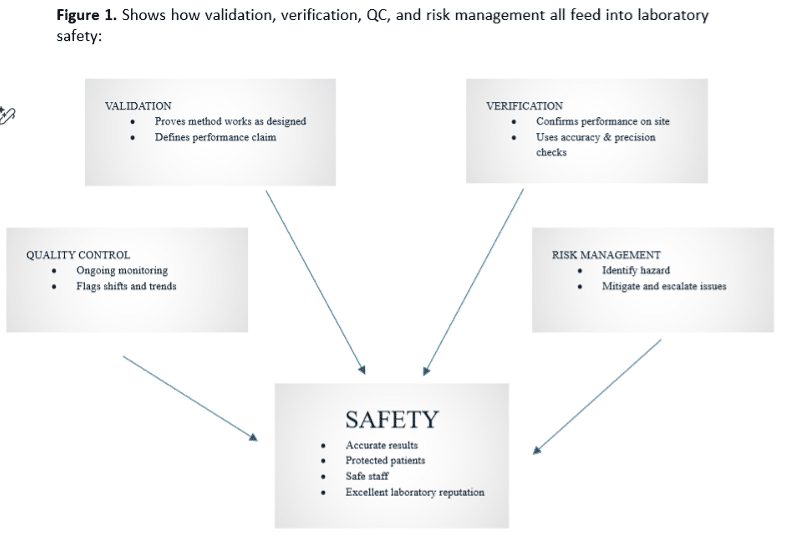
The article explains how validation, verification, quality control, and risk management work together to ensure clinical laboratory safety. Validation confirms a method’s reliability; verification ensures it performs correctly in a specific lab; QC detects ongoing errors; and risk management strengthens procedures—creating a comprehensive system that protects patients, staff, and testing accuracy.
A Critical Approach in Clinical Laboratories
Christian Baguistan, DCLS, MLS(ASCPi)CM, Volunteer Contributor

What Validation Really Delivers (Design Phase Safety)
Validation proves that a method or instrument operates as intended under all anticipated test conditions. Accuracy, precision, analytical sensitivity and specificity, reportable range/linearity, reference intervals (where applicable), and interference studies provide the comprehensive, evidence-based confirmation of a method’s performance. By precisely specifying or testing these properties before clinical use, validation avoids dangerous outcomes, such as false negatives that delay important treatment or false positives that lead to unnecessary or detrimental actions.
Limitations (such as hemolysis, sensitivity, cross-reactivity, and matrix effects) that are necessary for safe application are also made clear by validation. When employees are aware of a method’s limitations, they can identify when results or outcomes are unacceptable and take the necessary action (repeat, reflex, or confirmatory testing). To put it briefly, validation serves as the safety gate during the design phase, ensuring that the test is suitable before any patient sample is tested or handled.
Verification: Making Sure it Works Here (Implementation Phase Safety)
Even the most well validated methods still need to be challenged or verified in your particular laboratory setting with your specific instruments, reagent lots, staff, and environmental factors. Verification confirms that the manufacturer’s validated claims hold true on-site. Typical performance characteristics verified include accuracy or method comparison against reference materials or a comparator method, repeatability (within-run precision), reproducibility (between-run/day precision), and confirmation of the reportable range.
The purpose of verification is to protect against or address local hazards such as instrument installation problems, calibration drift, reagent handling, and workflow changes. It’s the implementation-phase safety gate, catching problems prior to routine reporting, so clinicians get reliable results from the start.
Quality Control as Continuation (Ongoing Safety)
Whenever verification is done and the testing is live, quality control (QC) continues the safety process. Performance of daily QC, external quality assessment or proficiency testing, as well as real time analytics (e.g., Westgard rules, moving averages) help identify random errors as well as shifts and trends that validation and/or verification cannot foresee. QC is the laboratory’s early-warning system. It keeps errors small, local, and correctable—before they become patient safety events.
Risk Management to Close the Loop (Anticipate, Mitigate, Escalate Issues on Safety)
Risk management is layered on top and includes mapping where failure happens in the testing process (pre-analytical, analytical, post-analytical), identifying hazards such as biological, chemical, and analytical risks, as well as validating procedures and policies (e.g., SOPs, training, competency assessment, etc.). Prioritizing mitigations and creating resilient workflows should be imperative and are made easier with the use of techniques such as root-cause analysis among others. Risk management closes the loop by ensuring that lessons learned from QC deviations, near misses, non-conforming events, and incident reports are applied to upgrade and update safer procedures.
How the Pieces Connect
- Validation proves the method is intrinsically safe to use (accurate and reliable by design).
- Verification confirms it is safe here and now (accurate and reliable in your lab).
- Quality Control ensures it stays safe (detects drifts and errors early).
- Risk Management makes the whole system robust (prevents, contains, and learns from failures).
Together, they protect patients (timely and correct diagnoses and treatments), staff (clear procedures, fewer surprises), and the organization (compliance, reputation, and cost control).
 Practical Steps to Tie Safety into Validation and Verification Processes
Practical Steps to Tie Safety into Validation and Verification Processes
- Write safety objectives into validation plans (e.g., define clinical decision thresholds and interference risks; specify acceptance criteria that reflect patient safety needs).
- Include environment and workflow-specific checks in verification (site calibration, reagent lot evaluation, sample stability under local transport/storage conditions, and chemical and hazard plans).
- Embed QC rules aligned to clinical risk (tighter controls near critical ranges; automatic reflex orders for high-impact results).
- Run periodic risk reviews (incident trend analysis, competency assessments, and root cause investigations) and update SOPs accordingly.
- Communicate clearly (validation summaries, verification reports, and QC dashboards) so clinicians understand method capabilities and limitations.
By making safety an explicit outcome of validation and verification—and sustaining it with QC and risk management—clinical laboratories create a reliable shield against errors, ensuring that every reported result supports the best possible patient care.
Christian Baguistan is a Clinical Laboratory Scientist Technical Specialist at Sutter Shared Laboratory in Livermore, California.
 Practical Steps to Tie Safety into Validation and Verification Processes
Practical Steps to Tie Safety into Validation and Verification Processes